TENS 7000 Digital TENS Unit with Accessories
User Manual

Chapter 1: INTRODUCTION
EXPLANATION OF PAIN
Pain is a warning system and the body ‘s method of telling us that something is wrong. Pain is important; without it abnormal conditions may go undetected, causing damage or injury to vital parts of our bodies.
Even though pain is a necessary warning signal of trauma or malfunction in the body, nature may have gone too far in its design. Aside from its value in diagnosis, long-lasting persistent pain serves no useful purpose. Pain does not begin until coded message travels to the brain where it is decoded, analyzed, and then reacted to. The pain message travels from the injured area along the small nerves leading to the spinal cord. Here the message is switched to different nerves that travel up the spinal cord to the brain. The pain message is then interpreted, referred back and the pain is felt.
EXPLANATION OF TENS
Transcutaneous Electrical Norvo Stimulation is a non-invasive, drugfree method of controlling pekin. TENS uses tiny electrical impulses sent through the skin to nerves to modify your pain perception. TENS does not cure any physiological problem; it only helps control the pain. TENS does not work for everyone; however, in most
patients it is effective in reducing or eliminating the pain, allowing for a return to normal activity.
HOW TENS WORKS
There is nothing “magic” about Transcutaneous Electrical Nerve Stimulation (TENS). TENS is intended to be used to relieve pain. The TENS unit sends comfortable impulses through the skin that stimulate the nerve (or nerves) in the treatment area. In many cases, this stimulation will greatly reduce or eliminate the pain sensation the patient feels. Pain relief varies by individual patient, mode selected for therapy, and the type of pain. In many patients, the reduction or elimination of pain lasts longer than the actual period of stimulation (sometimes as much as three to four times longer). In others, pain is only modified while stimulation actually occurs. You may discuss this with your physician or therapist.
IMPORTANT SAFETY INFORMATION!
Read instruction manual before operation. Be sure to comply with all “CAUTIONS” and “WARNINGS” in the manual. Failure to follow instructions can cause harm to user or device.
INDICATIONS FOR USE
This device is designed to be used for temporary relief of pain associated with sore and aching muscles in the shoulder, waist, back, upper extremities (arm) and lower extremities (leg) due to strain from exercise or normal activities.
Chapter 2: CAUTIONS
- Federal law allows over the counter (no prescription) sales of this device.
- Do not use this device for undiagnosed pain syndromes until consulting a physician.
- Patients with an implanted electronic device, such as a cardiac pacemaker, implanted defibrillator, or any other metallic or electronic device should not undergo TENS treatment without first consulting a doctor.
- Patients with heart disease, epilepsy, cancer or any other health condition should not undergo TENS treatment without first consulting a physician.
- Stimulation delivered by this device may be sufficient to cause electrocution. Electrical current of this magnitude must not flow through the thorax or across the chest because ii may cause a cardiac arrhythmia.
- Do not place electrodes on the front of the throat as spasm of the Laryngeal and Pharyngeal muscle may occur. Stimulation over the carotid sinus (neck region) may close the airways, make breathing difficult, and may have adverse effects on the heart rhythm or blood pressure.
- Do not place electrodes on your head or at any sites that may cause the electrical current to flow transcerebrally (through the head).
- This device should not be used While driving, operating machinery, close to water, or during any activity in which involuntary muscle contractions may put the user at undue risk of injury.
- Turn the TENS off before applying or removing electrodes.
- Isolated cases of skin irritation may occur at the site of electrode placement following long term application. If this occurs, discontinue use and consult your physician.
- If TENS therapy becomes ineffective or unpleasant, stimulation should be discontinued until its use is re-evaluated by a physician.
- Keep this device out of the reach of children.
- TENS 7000 devices have no AP/APG protection. Do not use ii in the presence of explosive atmosphere and flammable mixture.
Chapter 3: WARNINGS
- TENS devices have no curative value.
- TENS is a symptomatic treatment and as such suppresses the sensation of pain, which would otherwise serve as a protective mechanism.
- TENS is not effective for pain of central origin. (This includes headache.)
- Electronic monitoring equipment (such as ECG monitors and ECG alarms) may not operate properly when TENS stimulation is in Use.
- Caution should be used in applying TENS to patients suspected of having heart disease. Further clinical data is needed to show there are no adverse results.
- Electrodes should not be placed over the eyes, in the mouth, or internally.
- Do not use while sleeping
- Do not use during pregnancy unless directed by your physician.
Chapter 4: CONTRAINDICATIONS
- Do not use TENS over the carotid sinus (neck) region.
- TENS devices can affect the operation of demand type cardiac pacemakers.
- Do not use the TENS device if you have heart disease without consulting your physician.
- Do not stimulate on the site that may cause current to now transcerebrally – (through the head).
- Do no apply TENS for undiagnosed pain syndromes until etiology is established.
Chapter 5: ADVERSE REACTIONS
Skin irritation and electrode burns arc potential adverse reactions. If skin irritation occurs, discontinue use and consult your physician.
Chapter 6: GENERAL DESCRIPTION
The TE NS 7000 is a battery operated pulse generator that sends electrical impulses through electrodes to the body and reaches the nerves causing pain. The device is provided with two controllable output channels, each independent of each other. An electrode pair can be connected to each output channel.
The electronics of the TENS 7000 create electrical impulses whose Intensity, duration, number per second and modulation may be altered
with the controls/switches. Press buttons are very easy to use and the large liquid crystal display showing the exact mode and values of parameters are very convenient for patients.
Chapter 7: CONSTRUCTION
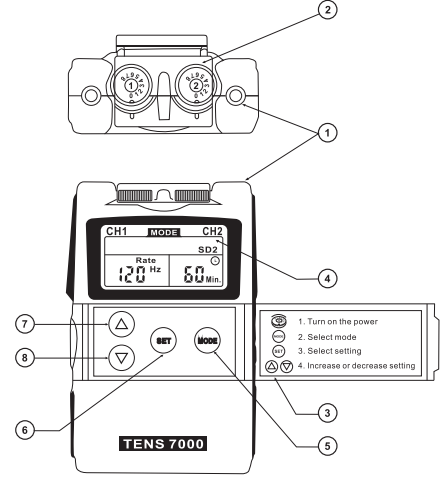
FRONT
- LEAD CONNECTOR
- INTENSITYCONTROL(ON/OFFSWITCH)
- PANELCOVER
- LIQUID CRYSTAL DISPLAY
- MODE CONTROL
- SET CONTROL
- INCREMENT CONTROL
- DECREMENT CONTROL
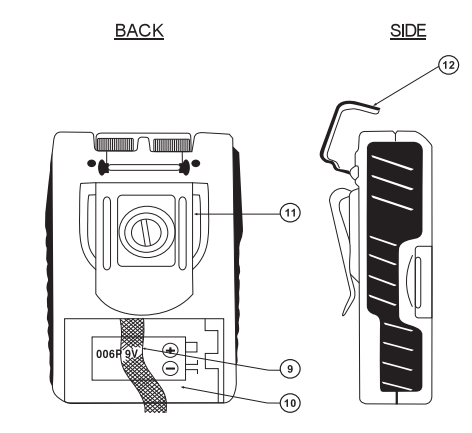
BACK
- (9) BATTERY STRIP
- (10) BATTERY CASE
- (11) BELTCLIP
SIDE
- (12) PROTECTIVE COVER
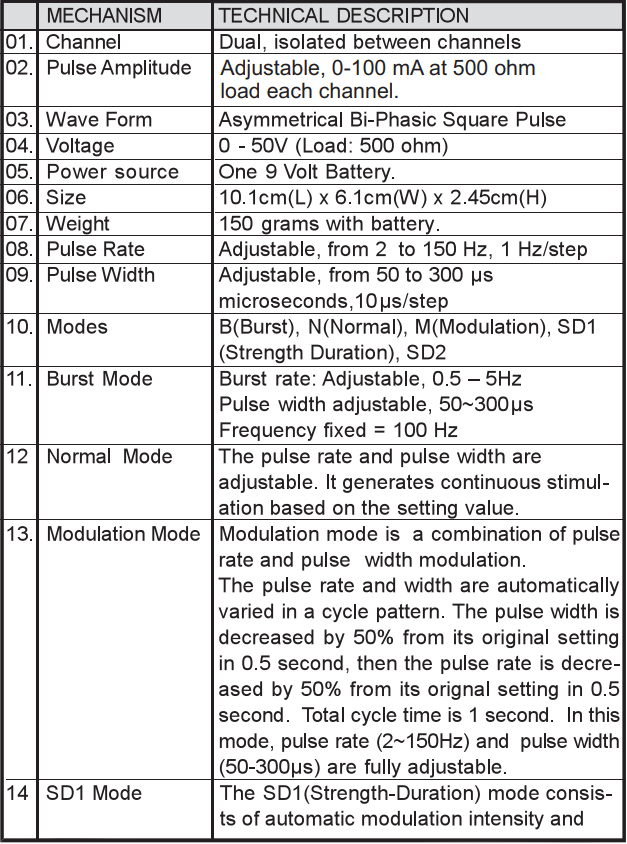
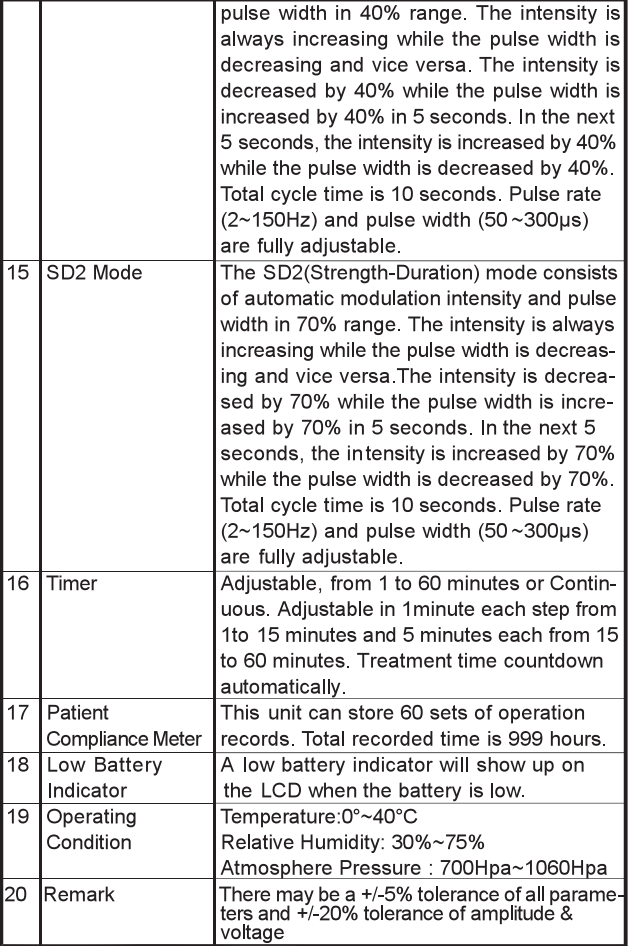
Chapter 8: TECHNICAL SPECIFICATIONS
The technical specification details of TENS 7000 are as follows:
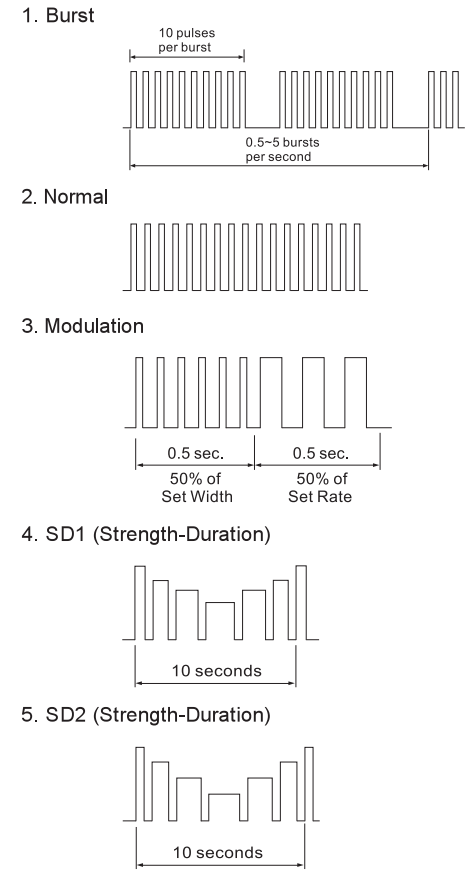
The waveforms of the 5 stimulation modes are as follows.
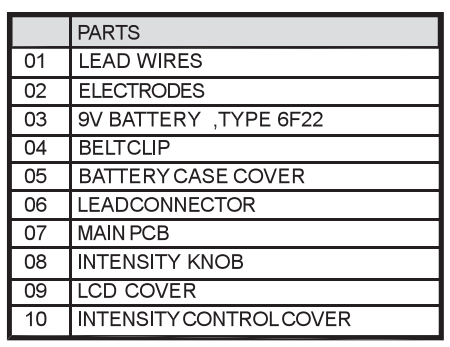
Chapter 9: REPLACEABLE PARTS
The replaceable parts and accessories of TENS 7000 devices are as given below:
Except leads, electrodes, battery and battery case cover, please do not try o replace the other parts of a device.
Chapter 10: ACCESSORIES
Each TENS 7000 comes complete with standard accessories and the standard labels as given below:

LABEL
 The label attached to the back of device contains important information about this device- model, supply voltage and caution. Please do not remove.
The label attached to the back of device contains important information about this device- model, supply voltage and caution. Please do not remove.
Chapter 11 : GRAPHIC SYMBOLS

Chapter 12: OPERATING INSTRUCTIONS
- Insert the 9V battery into the TENS 7000’s battery compartment. Make sure to remove the plastic seal on the 9V battery. Line up the positive and negative terminals on the battery with their corresponding terminals in the TENS 7000. Make sure that both Intensity control (ON/OFF Switch) knobs are in the off position.
- Insert the lead wires into the lead wire sockets on top of the TENS 7000.
- Open the electrode package. Then insert each lead wire pin into the pig tail of the electrodes.
- Place electrodes on your body surrounding or directly on the area of pain.
- Slowly turn on the TENS 7000 by rotating the Intensity control (ON / OFF Switch) knobs.
- Select the mode and settings you want.
- Slowly increase or decrease the intensity by rotating the intensity control (ONN/OFF) knob clockwise to increase or counter clockwise to decrease.
- After treatment, turn the TE NS 7000 off by rotating the Intensity control (ON/OFF Switch) counter clockwise to the zero setting.
Chapter 13 : PARAMETER CONTROLS
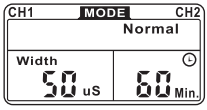
PULSE WIDTH (DURATION)
Pulse width is how wide each pulse is. Typically, the higher the pulse width the stronger the stimulation feels. If set high enough, sometimes it will produce a contraction which is typically not what you want to elicit with a TENS unit. Various pulse widths are capable of stimulating different groups of nerve fibers.
PULSE RATE
Pulse rate is also known as pulses per second. It is the number of times a pulse occurs in one second. This setting can be adjusted to excite certain nerves to overcome accommodation effects and will feel the sensation of steady continuous stimulation.
TREATMENT MODE
Normal or Conventional TENS offers the practitioners complete control over all !he various treatment parameters of the instrument.
Burst Mode is analogous lo the Low Rale TENS technique except the low frequency individual pulses are replaced by individual “bursts” of 7-10 individual pulses. II is thus a combination of Conventional TENS and Low Rate TENS. In Burst Mode, the treatment frequency is fixed by !he instrument and is not adjustable with the Frequency Rate control.
Modulated Mode attempts lo prevent nerve accommodation by continuously cycling the treatment intensity. When using Modulated Mode increase the intensity only when the unit is al the maximum intensity of the modulation cycle. If the intensely is increased during a low intensity period of the modulation cycle, the patient should increase !he intensely slowly until the modulation cycle reaches !he maximum lo insure a true maximum intensity output.
Strength – Duration Modulation (SD1 & SD2) consists of alternating modulated amplitude and width so that one parameter is always decreasing while the other is increasing and vice versa. The amplitude decreases from the amplitude control selling and returns lo the selling. The width decreases from the width control selling and returns to the selling.
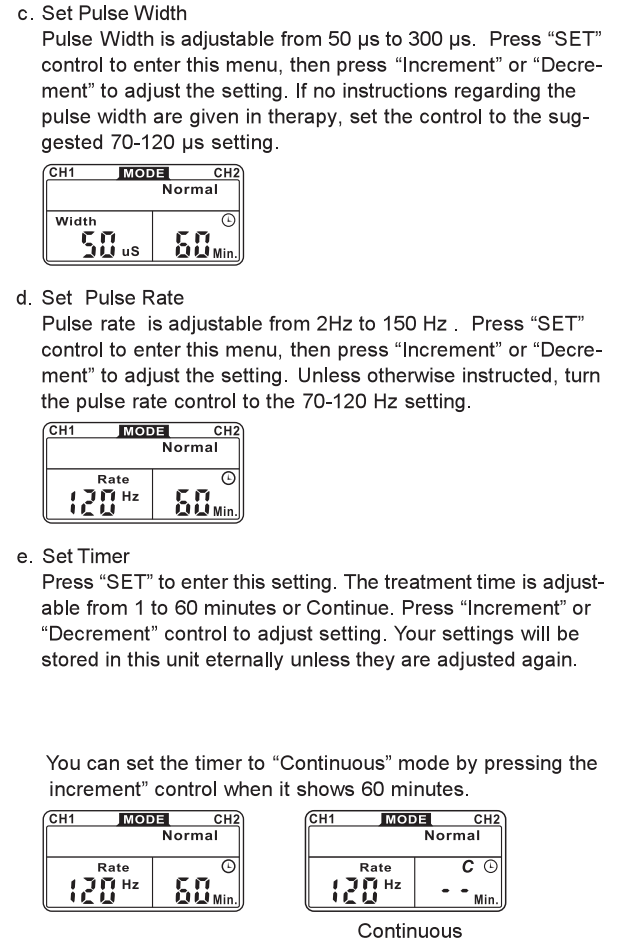
TIME DURATION
The onset of pain relief should occur shortly after the intensely selling has been determined. However, in some cases, pain relief may lake as long as 30 minutes lo achieve. TENS units are typically operated for long periods of lime, with a minimum of 20 30 minutes and in some post-operation protocols, as long as 36 hours.
In general, pain relief will diminish within 30 minutes of the cessation of stimulation.
Chapter 14: ATTACHMENT OF ELECTRODE LEAD WIRES
The wires provided with the system insert into the jack sockets locale on lop of the device. Holding the insulated portion of the connector, push the plug end of !he wire into one of !he jacks (see drawing); one or two sets of wires may be used.
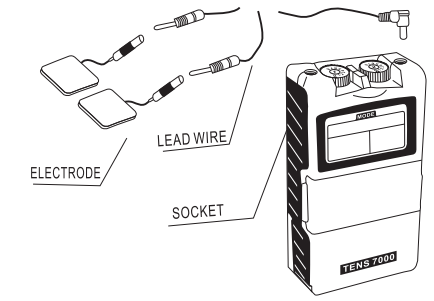
After connecting the wires lo the simulator, Allah each wire to an electrode. Use care when you plug and unplug the wires. Jerking the wire instead of holding the insulated connector body may cause wire breakage.
CAUTION
Do not insert the plug of the patient lead wire into any AC power supply socket.
Chapter 15: LEAD WIRE MAINTENANCE
Clean the wires by wiping with a damp cloth. Coating them lightly with talcum powder will reduce tangling and prolong life.
Chapter 16: ELECTRODE OPTIONS
The electrodes are disposable and should be routinely replaced when they start to lose their adhesive nature. If you are unsure of your electrode adhesive properties, order replacement electrodes. Replacement electrodes should be approved for over-the-counter use. Follow application procedures outlines in the electrode packaging to maintain optimal stimulation and to prevent skin irritation.
Chapter 17: ELECTRODE PLACEMENT
The placement of electrodes can be one of the most important parameters in achieving success with TENS therapy. Placing the electrodes at least 2″ apart but no more than 6″ apart will help with successful therapy.
Every patient responds to electrical stimulation differently and their needs may vary from the conventional settings suggested here. If the initial results are not positive, speak to your physician about alternative stimulation settings and/or electrode placements. Once an acceptable placement has boon achieved, mark down the electrodes sites and the settings, so the patient can easily continue treatment at home.
CONTIGUOUS PLACEMENT
This is the most common placement technique. It involve placing the electrodes alongside the area of localized pain site, in such a way as to direct the flow of current through or around the area of pain.
In a single channel application, this would involve placing each pad on either side of the pain site if the pain is localized on a limb and deep within the tissue. Pad placement on the posterior and anterior aspects of the affected limb will allow the current to flow completely through the limb and thus through the endogenous pain site.
With a two channel application, may either direct the flow to cross through the pain site or, in what is called the “bracket” method allowing the current flow on either side of the painful area, generally through the nerve branches that feed into the pain site.
Chapter 18: TIPS FOR SKIN CARE
To avoid skin irritation, especially if you have sensitive skin, follow these suggestions:
- Wash the area of skin where you will be placing the electrodes, using mild soap and water before applying electrodes, and after taking them off. Be sure to rinse soap off thoroughly and dry skin well.
- Excess hair may be clipped with scissors; do not shave stimulation area.
- Wipe the area with skin preparation wipes. Let the area dry. Apply electrodes as directed.
- Many skin problems arise from the “pulling entrée” from adhesive patches that are excessively stretched across the skin during application. To prevent thief, apply electrode from center outward; avoid stretching over the skin.
- To minimize “pulling egress”, tape extra length of lead wires to the skin in a loop to prevent tugging on electrodes.
- When removing electrodes, always remove by pulling in the direction of hair growth.
- It may be helpful to rub skin lotion on electrode placement area when not wearing electrodes.
- Never apply electrodes over irritated or broken skin.
Chapter 19: APPLICATION OF RE-USABLE SELF ADHESIVE ELECTRODES
Application
- Clean and dry !he skin al !he prescribed area thoroughly will soap and walker prior lo application of electrodes.
- Insert !he lead wire into !he pin connector on !he pre-wired electrodes.
- Remove the electrodes from the protective liner and apply the electrodes firmly to the lrealmenl 1ite. Make sure that !he unit is turned off prior lo applying !he electrodes.
Removal
- Turn off !he unit! prior lo removing !he electrodes.
- Lidl at !he edge of electrodes and peel; do not pull on the lead wires because ii may damage the electrodes.
- Place the electrodes on the liner and remove !he lead wire by wasting and pulling al !he same lime
Care and Storage
- Between uses, store the electrodes in the resealable bag in a cool dry place.
- It may be helpful lo improve repealed application by spreading a few drops of cold walker over !he adhesive and !urn !he surface up to air dry. Over Saturation with water will reduce the adhesive properties.
Important
- Do no! apply lo broken skin.
- The electrodes should be discarded and re-ordered from your physician when they are no longer adhering.
- The electrodes are intended for single patient use only.
- If irrigation occurs, discontinue use and consult your physician.
- Read !he instructions for use of self-adhesive electrodes before application.
Chapter 20: ADJUSTING THE CONTROLS
- Panel Cover:
A lid covers !he controls for selecting mode and adjusting selling’s. Your medical professional may wish lo set these controls for you and request that you leave the cover in place.

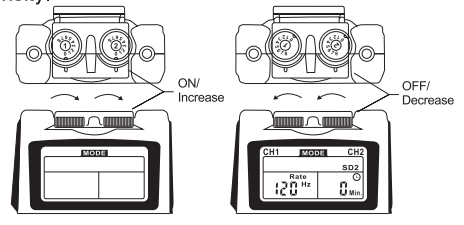
- Power On/Off Switch and Intensity Controls:
If both controls are in the off-position, the device is switched off.
By turning !he controls clockwise, !he appropriate channel is switched on and the indicator of power (CH1 or CH2) will reveal on the LCD.
The current strength of the impulses transmitted to the electrodes increases further when the control is turned clockwise.
To reduce !he current strength or switch The device off, !urn The
control counter clockwise to the required setting or off-position, respectively.
The controls are protected by a cap lo avoid unintentional change
of intensity.

- Lead Connector
Connection of the electrodes is made with the Two lead wires. The device must be switched off before connecting the cables. Both intensely controls must be al the Off position. Electrodes must be pressed firmly on The skin.

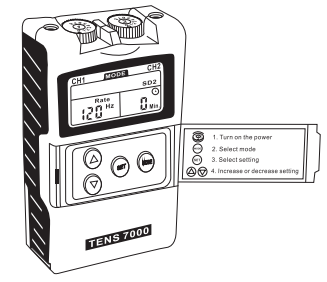
- Mode Control (MODE)
There are 5 modes available – Burst, Normal, Modulation, SD1 and SD2. The mode can be selected by pressing the “MODE” control. - Set Control (SET)
By pressing the “SET” control, you may enter the setting you intend to make adjustment. You may start to set the value by pressing the “Increment” and “Decrement” controls when the value is flashing. - Increment Control (▲)
This bullion controls the increase of selling’s. When pressing this button, the parameter will increase. - Decrement Control (▼)
This button controls the decrease of parameter. When pressing this bullion, !he parameter will decrease. - Timer
The unit has a lamer of 1-60 minutes and Continue. It can be adjusted by pressing the “Sell” and “Increment” or “Decrement” controls. The lrealmenl lime will countdown automatically one minute by one minute. Its outpull will be shul off when lime is up. - Low Battery Indicator

A low battery indicator will show up on !he liquid crystal display when the battery need lo be replaced as soon as possible. The unit may continue lo operate for a few more hours depends on the selling intensity level. - Steps lo Set a New Program
The selling’s can be adjusted according lo the following steps.
a. Turn on the Intensity
After the electrodes are placed firmly on skin and the lead wires are plugged in the socket of device, turn the on/off control clockwise. The liquid crystal display will be light up.
b. Select a Mode
Select a mode by pressing !he “MODE” control. The mode you select will show up on the lop of liquid crystal display. There are 5 modes of your option, including -Burst, Normal, Modulation, SD1 and SD2.

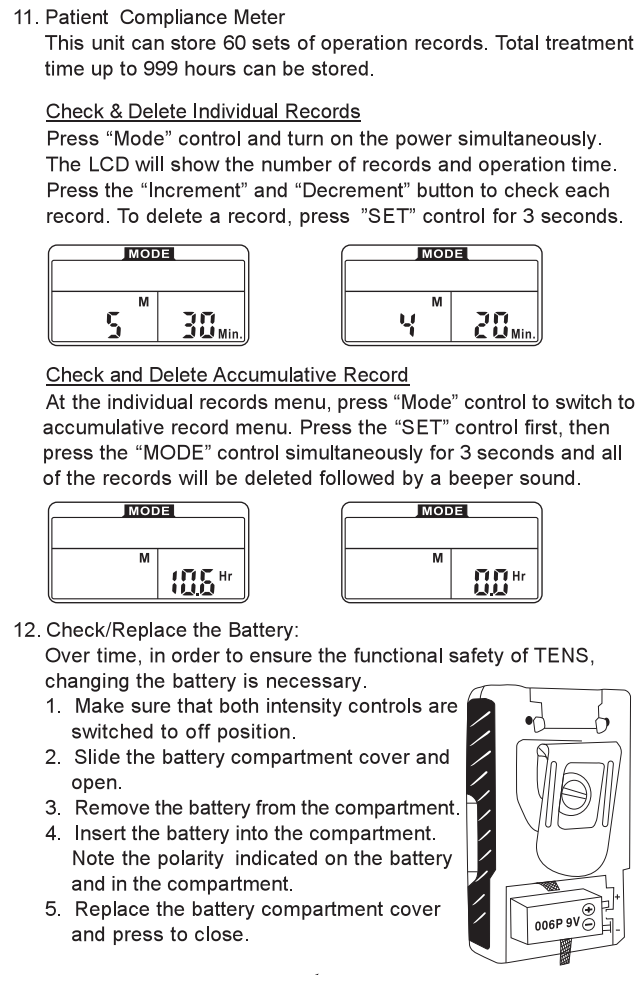
Chapter 21: BATTERY INFORMATION
PRECATIONS
- Remove battery if equipment is not likely to be used for some time.
- Please recycle the used battery in accordance with domestic regulation.
- Do not throw !he used battery into fire.
If you use rechargeable batteries, please follow !he instructions.
RECHARGEABLE BATTERIES (NOT INCLUDED)
Prior to !he use of a new unit!, the rechargeable battery should be charged according to !he battery manufacturer’s instructions. Before using the battery charger, read all instructions and cautionary markings on the battery and in this instruction manual.
After being stored for 60 days or more, !he batteries may lose their charge. After long periods of storage, batteries should be charged prior lo use.
BATTERY CHARGING
- Plug !he charger into any working 110 or 220/240Vmains electrical outlet The use of any attachment not supplied with the charger may result in the risk of fire, electric shock, or injury lo persons.
- Follow !he battery manufacturer’s instructions for charging lime.
- After the battery manufacturer’s recommended charging time has been completed, unplug !he charger and remove the battery.
- Batteries should always be stored in a fully charged stale. To ensure optimum battery performance, follow these guidelines:
(a) Although overcharging the batteries for up to 24 hours will no! damage them, repealed overcharging may decrease useful battery life.
(b) Always store batteries in their charged condition. After a battery has been discharged, recharge ii as soon as possible. If the battery is stored more than 60 days, ii may need to be recharged.
(c) Do not short !he terminals of !he battery. This will cause the battery to get hot and can cause permanent damage. Avoid storing the batteries in your pocket or purse where !he terminals may accidentally come into contact with coins, keys or any metal objects.
(d) WARNINGS:
1. Do no! attempt lo charge any other types of batteries in your charger, other than rechargeable batteries made for your charger. other types of batteries may leak or burst
2. Do not incinerate !he rechargeable battery as it may explode!
Chapter 22: MAINTENANCE, TRANSPORTATION AND STORAGE OF THE DEVICE
- Non-flammable cleaning solution is suitable for cleaning the device.
Note: Do no! smoke or work with open lights (for example, candles, etc.) when working with flammable liquids. - Stains and spots can be removed with a cleaning agent
- Do not submerge the device in liquids or expose it to large amounts of water.
- Return !he device to !he carrying box with sponge foam lo ensure that the unit is well-precelled before transportation.
- if the device is no! lo be used for a long period of time, remove the batteries from !he battery compartment (acid may leak from used batteries and damage the device). Put the device and accessories in carrying box and keep it in cool dry place.
- The packed TENS device should be stored and transported under !he temperature range of -20°C ~ + 60°C, relative humidity 20% – 95%, atmosphere pressure 500 hPa – 1060 hPa.
Chapter 23: SAFETY-TECHNICAL CONTROLS
For safety reasons, review !he following checklist before using your TENS 7000,
- Check the device for external damage, deformation of the housing, damaged or defective output sockets,
- Check !he device for defective operating elements, legibility of inscriptions and labels, make sure the inscriptions and labels are not distorted.
- Check !he usability of accessories, patient cable undamaged, electrodes undamaged. Battery is no! corroded
Please consult your distributor if !here are any problems with device and accessories,
Chapter 24: MALFUNCTIONS
Should any malfunctions occur while using !he TENS 7000, check
– whether the parameters are set lo the appropriate form of Therapy.
Adjust !he control correctly,
– whether the cable is correctly connected lo the device, The cables should be inserted completely into the sockets.
– whether the LCD reveals the menu, If necessary, insert a new battery,
for possible damage lo the cable, Change the cable if any damage is detected,
- If there is any other problem, please return the device to your distributor. Do not try to repair a defective device.
Chapter 25: CONFORMITY TO SAFETY STANDARDS
The TENS 7000 devices are in compliance with IEC 60601-1:2005.
Chapter 26: WARRANTY
All TENS 7000 models carry a warranty of one year from the dale of delivery, The warranty applies to the stimulator only and covers both parts and labor relating thereto,
The warranty does not apply lo damage resulting from failure to follow the operating instructions, accidents, abuse, alteration or disassembly by unauthorized personnel.

Manufactured for:
Roscoe Medical Inc.
6753 Engle Road
Middleburg Heights, OH 44130
Phone: (800) 376-7263
www.roscoemedical.com
42-DT7202_01